Navigating the intricacies of cellular proteins
Ever wish there were a way to take a snapshot of two proteins as they interact or remain unbound? Now, the door is open to study protein interplay and functional states while keeping the structural integrity of cells. Our innovation Naveni® TriFlex Technology uncovers protein complex formation and disassembly in response to different stimuli in situ. This revolutionary proximity-based technology concurrently visualizes two proteins in a free and complex state, within any cell compartment, and enables you to shed light on the hidden lives of proteins. The technology, which is a development of our Naveni® in situ proximity ligation technology, is built into the product Naveni® TriFlex Cell – a kit with the flexibility to be combined with your own primary antibodies.
Naveni® TriFlex Cell relies on two user-determined primary antibodies against the targets of interest, and on proprietary TriFlex Navenibodies (in orange), which are antibody-based proximity reagents. Only proteins located at <40 nm distance are recorded as interacting. Naveni® TriFlex detects total protein A (i.e., both free and in complex with B, yellow), total protein B (maroon), and the AB interaction (blue). The detected A, B and AB signals are amplified and generate fluorescent readout in three channels corresponding to each protein pool.
From the cradle to the grave
Naveni® TriFlex Cell was used to simultaneously detect free and interacting Lamin B1 and Histone H3
Birth: In mitotic cells, Lamin B signals (yellow) are diffuse due to nuclear envelope breakdown, whereas Histone H3 (magenta) remains around the metaphase plate. Lamin B/Histone H3 complexes (cyan) have disassembled.
Life: In non-dividing cells, free Lamin B is localized in the nuclear envelope, and multiple Lamin/Histone H3 interactions are observed in the nucleoplasm together with chromatin-bound Histone H3.
Death: During the initial stages of oncosis, the dying cell’s nucleus swells and the nucleoplasm contains less densely packed complexes along with unbound Lamin B and Histone H3.
Highly abundant proteins visualize cell-cell adhesion
Naveni® TriFlex Cell was used to detect the interaction of β-catenin/ E-cadherin, as well as total β-catenin and E-cadherin. Images depict what is seen in each of the three different channels, as well as a merged image. β -catenin and E-cadherin are highly expressed proteins and the interaction between the two proteins is important for cell-cell adhesion.
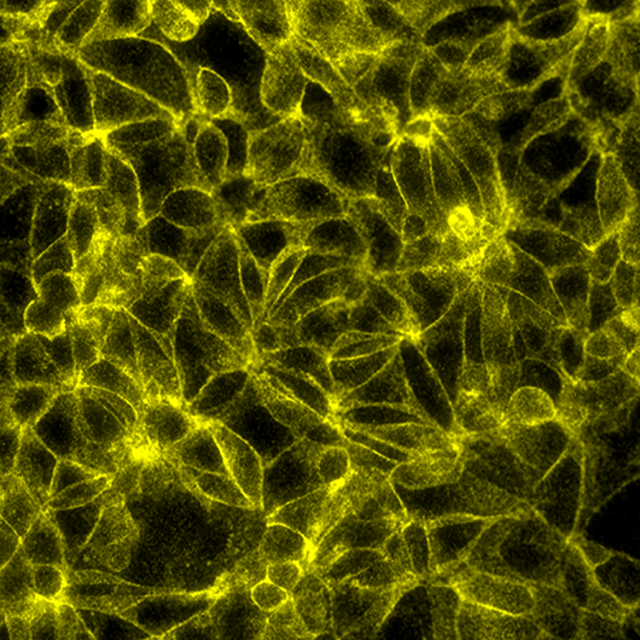
Total protein signal of Beta-catenin in MCF7 cells, signals visualized in FITC filter set (yellow)

Total protein signal of E-Cadherin in the same MCF7 cells, signals visualized in Cy3 filter set (magenta)

Beta-catenin/E-Cadherin interaction in the same MCF7 cells, signals visualized in Cy5 filter set (cyan)

Merge of the three filter sets, Beta-catenin signals in yellow and E-Cadherin signals in magenta. Beta-catenin/E-Cadherin interaction in cyan. Overlapping signal is white.
Overlap of fluorescent signals is not a synonym for protein interaction
What appears to be an overlap of two fluorescent signals needs to be distinguished from protein-protein interaction. Learn how and why below.
Naveni® TriFlex Cell is used to simultaneously detect two target proteins and their interaction. In the example below, the protein targets reside in separate cellular compartments and are not expected to interact. In line with this, the mitochondrial protein COX1 (FITC, in yellow) and the Golgi apparatus protein GM130 (Cy3, in magenta) are clearly detected in their respective compartments but no proximity signal is generated (Cy5, in cyan). Notably, the proteins appear to be present in the same physical structure in the cell – there is an overlap of fluorescence when channels are merged. In traditional immunofluorescence, this could falsely be interpreted as co-localization and interacting proteins.

Total protein signal of COX1 in MCF7 cells, signals visualized in FITC filter set (yellow), nuclei shown in blue.

Total protein signal of GM130 in the same MCF7 cells, signals visualized in Cy3 filter set (magenta), nuclei shown in blue.
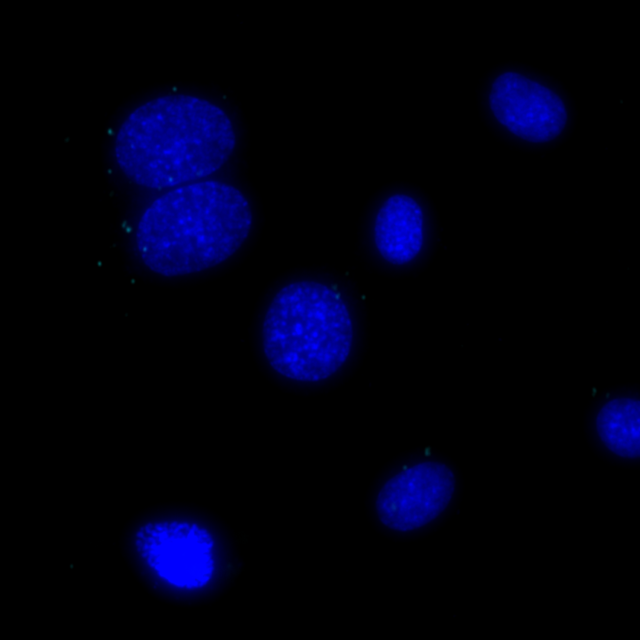
Undetectable signals for COX1/GM130 interaction in the same MCF7 cells, signals visualized in Cy5 filter set (cyan), nuclei shown in blue.

Merge of the three filter sets, COX1 signals in yellow and GM130 signals in magenta. Where COX1 and GM130 signals overlap, the signal is white and could falsely be interpreted as interaction.
How to detect free and interacting proteins
Use or flexible product Naveni® TriFlex Cell. If you are only interested in one protein-protein interaction, we recommend NaveniFlex Cell.
How to detect free and interacting proteins
Use or flexible product Naveni® TriFlex Cell. If you are only interested in one protein-protein interaction, we recommend NaveniFlex Cell.

Naveni® TriFlex Cell
The kit enables fluorescent staining of protein-protein interactions, concurrently visualizing the two proteins in their free state.
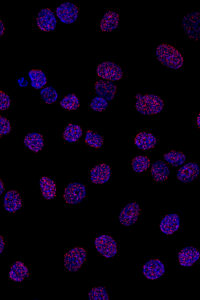
NaveniFlex Cell
The assay is designed to be used with a mouse and rabbit primary antibody pair. Detection with TEX615 fluorophore is included.
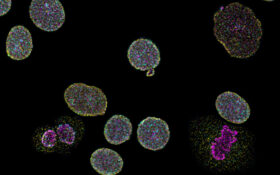
Naveni® TriFlex Cell
The kit enables fluorescent staining of protein-protein interactions, concurrently visualizing the two proteins in their free state.
NaveniFlex Cell
The assay is designed to be used with a mouse and rabbit primary antibody pair. Detection with TEX615 fluorophore is included.
Expert opinion:
’The TriFlex product makes it possible to measure protein co-location with high precision, independent of microscope resolution.’
This is what imaging expert Carolina Wählby, professor in quantitative microscopy at the Dept. of Information Technology and SciLifeLab, and main contributor to the development of image analysis software CellProfiler, had to say about our newest technology.


